The European virus archive (EVA) is an international non-profit association (AISBL) under the Belgian Law created in March 2025.
EVA is a virology consortium providing the largest collection of viral resources worldwide, with the mission of facilitating virology research and protecting Public Health. By providing academic and private virology researchers with the necessary resources to study viruses and address emerging global threats, EVA addresses critical gaps in pandemic preparedness and response: No central virus collection exists in Europe and there is a need for standardised research using reference material and standardised services for reproducibility in translational research.
Becoming EVA AISBL
Before being an Association, the European Virus Archive benefited from the support of the European Commission through three different grants over the last 15 years:
- EVA (2009-2014), 7th Framework Programme for Research under grant agreement ID 228292
- EVA goes global (2014-2019), Horizon 2020 - Research and innovation under grant agreement ID 653316
- EVA-GLOBAL (2020-2024), Horizon 2020 - Research and innovation under grant agreement ID 871029
EVA AISBL’s Missions
- Gather leading expertise for the collection, amplification, characterisation, standardisation and authentification of human, animal and plant virus resources.
- Develop state-of-the-art technologies for the production of virus-derived products and non-infectious materials for use in diagnostics and research.
- Facilitate access to expertise, resources and services for researchers in academia and industry.
- Support Public Health response and research during viral outbreaks.
EVA AISBL’s Collection
- “One virology concept”: large, diverse and “Fit for Purpose” collection
- Common quality grading and QMS (towards ISO 20387and ISO 13485) and Compliance with Nagoya Protocol
- Easy access to the collection: Online ordering and evaluation, Customer support, ratified MTA, Logistics procedures.
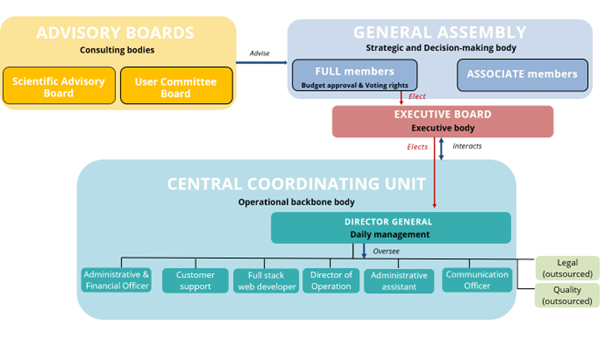
EVA AISBL’s Governance
- General Assembly: Decision-making body
- Executive Board: Managing and executive body
- Director General: Ensures the daily management
- Central Coordinating Unit: Ensures the scientific follow-up, coordinate the activity programs, the daily administration and assists the DG
- Advisory Committees: Provides scientific, ethical, and strategic guidance upon decision of the General Assembly
Documents:
-
Download our Legal Statutes :
 EVA-AISBL statutes
EVA-AISBL statutes
-
Download our Gender Equality Plan:
 EVA-AISBL Gender Equality Plan
EVA-AISBL Gender Equality Plan
- Download our 2025 Annual Report (Incomming)
Members of the EVA Executive Board
- Hervé BOURHY (Institut Pasteur, France)
- Tatjana AVSIC ZUPANC (Univerza v Ljubljani, Slovenia
- Rémi CHARREL (Aix-Marseille Université, France)
- Boris KLEMPA (Biomedicínske centrum Slovenskej akadémie vied, Slovakia)
- Amber Hartman SCHOLZ (Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH, Germany)
- Maria Beatrice BONIOTTI (Istituto Zooprofilattico Sperimentale della Lombardia ed Emilia Romagna, Italy)
Director General:
- Bruno COUTARD (Aix-Marseille Université, France)
Governance structure
To contact us:
Access and benefit sharing under the Convention on Biological Diversity and the Nagoya Protocol
The EVA-AISBL community supports fair and equitable benefit sharing as conceived in various United Nations frameworks. The consortium is actively implementing a CBD/Nagoya Protocol compliance strategy.
Privacy policy and GDPR compliance
Personal data is collected when you create an account and when you place an enquiry for any product or service, or when you initiate these processes. Personal data is collected to allow you to personalise your use of the EVA-AISBL website and related services’ (placing enquiries, email notifications, newsletters) and for the delivery of ordered products...
Medias used on this website
Credits
This transmission electron micrograph (TEM) revealed some of the ultrastructural morphology of a number of intact rotavirus double-shelled particles. Distinctive rim of radiating capsomeres.- 1981-
This negative stained transmission electron micrograph (TEM) shows recreated 1918 influenza virions that were collected from supernatants of 1918-infected Madin-Darby Canine Kidney (MDCK) cells cultures 18 hours after infection.
To separate these virions, the MDCK cells are spun down (centrifugation), and the 1918 virus in the fluid is immediately fixed for negative staining. The solid mass in lower center contains MDCK cell debris that did not spin down during the procedure.
Transmission electron micrograph, negative stain image of the polio virus (1971).
Created by CDC microbiologist Frederick A. Murphy, this colorized transmission electron micrograph (TEM) revealed some of the ultrastructural morphology displayed by an Ebola virus virion.
This image was created by Nahid Bhadelia, M.D., Assistant Professor of Medicine in the Section of Infectious Diseases, and the Associate Hospital Epidemiologist at Boston Medical Center. It was captured while the U.S. Centers for Disease Control and Prevention’s (CDC) 2014 Domestic Ebola ETU Training Course for healthcare workers was underway. The program had been designed in order to educate participants who would be deployed as members of the West African Ebola Response team, as to the proper protocols to be followed when treating Ebola hemorrhagic fever (Ebola HF) patients. Here you can see two participants who’d paired up in order to dress in their required personal protective equipment (PPE). By applying the buddy system, each healthcare worker can act as the secondary check for the other, thereby, assuring that the each other’s PPE had been properly secured.
This transmission electron micrograph (TEM) revealed some of the ultrastructural morphology of a number of intact rotavirus double-shelled particles. Distinctive rim of radiating capsomeres.- 1981-
This negative stained transmission electron micrograph (TEM) shows recreated 1918 influenza virions that were collected from supernatants of 1918-infected Madin-Darby Canine Kidney (MDCK) cells cultures 18 hours after infection.
To separate these virions, the MDCK cells are spun down (centrifugation), and the 1918 virus in the fluid is immediately fixed for negative staining. The solid mass in lower center contains MDCK cell debris that did not spin down during the procedure.
Transmission electron micrograph, negative stain image of the polio virus (1971).
Created by CDC microbiologist Frederick A. Murphy, this colorized transmission electron micrograph (TEM) revealed some of the ultrastructural morphology displayed by an Ebola virus virion.
This image was created by Nahid Bhadelia, M.D., Assistant Professor of Medicine in the Section of Infectious Diseases, and the Associate Hospital Epidemiologist at Boston Medical Center. It was captured while the U.S. Centers for Disease Control and Prevention’s (CDC) 2014 Domestic Ebola ETU Training Course for healthcare workers was underway. The program had been designed in order to educate participants who would be deployed as members of the West African Ebola Response team, as to the proper protocols to be followed when treating Ebola hemorrhagic fever (Ebola HF) patients. Here you can see two participants who’d paired up in order to dress in their required personal protective equipment (PPE). By applying the buddy system, each healthcare worker can act as the secondary check for the other, thereby, assuring that the each other’s PPE had been properly secured.
 Rotavirus double-shelled particles
Rotavirus double-shelled particles Influenza virions (TEM)
Influenza virions (TEM) Poliovirus (TEM)
Poliovirus (TEM) Ebola hemorrhagic fever
Ebola hemorrhagic fever CDC 2014 Domestic Ebola ETU Training Course for healthcare workers
CDC 2014 Domestic Ebola ETU Training Course for healthcare workers